
Forįurther information, consult your state's handbook of Science Safety. 
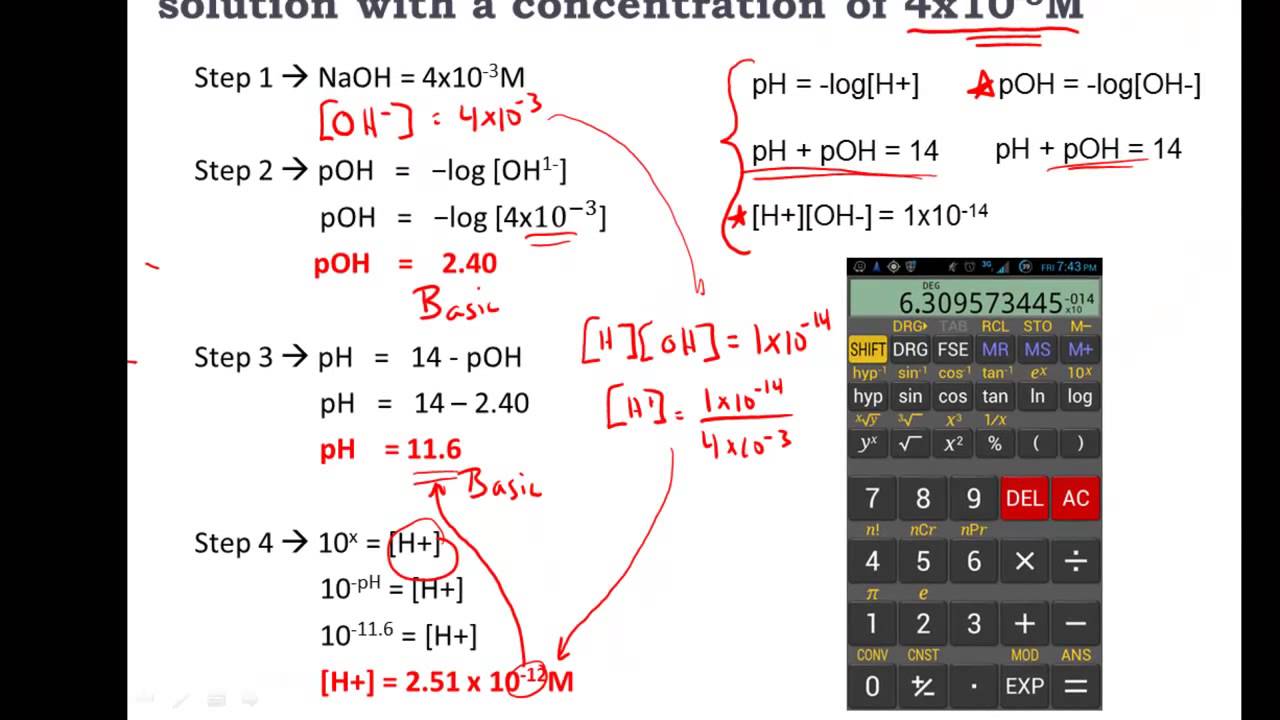
Materials used in a project is the sole responsibility of each individual. Reading and following the safety precautions of all Should be undertaken only in appropriate settings and with appropriate parental Implementation of any Science Project Idea Warning is hereby given that not all Project Ideas are appropriate for all In addition, yourĪccess to 's website and Science Fair Project Ideas is covered byĮ's Privacy Policy and site Terms of Use, which include limitations Renounce any claims against that arise thereof. If your guessed pH is < 7 the H+ concentration will be higher and you will have to add protons to establish that level and neutralize the 10-7 concentration at pH 7. By accessing the Science Fair Project Ideas, you waive and Calculate the concentration of OH- and H+ at that pH and determine the number of protons that you would have to add (or subtract) to your volume of pure water to reach that pH. are simply listed as > 1 which indicates that they dissociate 100 when placed in water. 7 is neutral- it is neither basic nor acidic and stands for distilled water. To calculate the pH of mixtures of acid and base solutions. The scale ranges from 0-14, with 0 being the most acidic and 14 being the most basic. Regarding the Science Fair Project Ideas and is not responsible or liable forĪny loss or damage, directly or indirectly, caused by your use of such pH is the way to measure how acidic or basic a solution is. does not make any guarantee or representation If it did does that necessarily mean it is water?Į provides the Science Fair Project Ideas for informational
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
